How to Find Tardigrades (Water Bears) in Your Own Backyard
 Aug 10, 2016 • 2:47 AM UTC
Aug 10, 2016 • 2:47 AM UTC United States
United States 140x Magnification
140x Magnification Microorganisms
Microorganisms
Ricky Cordova
Led by a small team of undergraduates, the Stanford Tardigrade Project is aimed at studying wondrous lives of the world's toughest creatures -- TARDIGRADES.
1posts
6comments
1locations

View in Media Gallery
Tardigrades, also known as Water Bears or Space Bears, are incredible members of the microcosmos that can be found just about anywhere in the world. These eight-legged invertebrates are only 0.1-1mm long, yet they are certainly the world’s toughest animals!

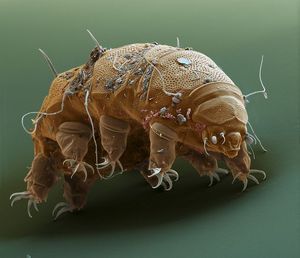
Just how tough are they? They have been shown to survive temperatures as low as -272°C and as high as 151°C (Rahm, 1923, 1924, 1926), pressures 6 times greater than those at the bottom of the deepest ocean trench on Earth(Seki & Toyoshima, 1998), over 1000 times the human lethal dose of heavy ion and gamma radiation (Horikawa et al. 2006), and more. In fact, tardigrades have even survived for weeks in space (Jönsson et al. 2008)!

View in Media Gallery
Typically considered aquatic animals, these small creatures require a film of water around their bodies to permit locomotion and gas exchange (Kinchin, 1994).

View in Media Gallery
Fortunately for the tardigrade, if there isn’t enough water, or environmental conditions become unfavorable, they can enter a state of “cryptobiosis” – a reversible suspension of metabolism (Keilin, 1959) – from which they can be revived decades later (Tsujimoto et al. 2016) when favorable conditions return. With the aid of some helpful, protective proteins, they release much of their body-water content, shrink into a “tun,” and remain in the same spot for as long as they need to.

View in Media Gallery
This cryptobiotic adaptation has allowed many species of tardigrades to thrive on land in mosses, colonies of lichen, and liverworts, where periodic drying and wetting is common (Ramazzotti & Maucci, 1983). These habitats also contain plenty of food for both herbivorous and carnivorous tardigrades: algae, cyanobacteria, rotifers, nematodes, and other tardigrades.


In this post, I hope to share a step-by-step guide to finding tardigrades in your own backyard. Because these amazing creatures comprise an entire phylum of animals (phylum Tardigrada), of which only about 1000 species have been described, there is even a chance that you’ll find tardigrades from an entirely new species, never before seen!
Note : This guide also works great for finding nematodes and rotifers, which are almost always found occupying the same habitats as the tardigrades.
***
Each morning on my way to work, I pass a short, wooden fence that is home to many colonies of lichen.
Note : This guide also works great for finding nematodes and rotifers, which are almost always found occupying the same habitats as the tardigrades.
***
Each morning on my way to work, I pass a short, wooden fence that is home to many colonies of lichen.


Every time I’ve seen it, I have wondered whether these lichen hold tardigrades in their leafy folds.



I thought this would be the perfect place to demonstrate how to look for tardigrades.
Here are the materials you will need:
Paper bags Razor blade or pocket knife (optional) Pen or Marker GPS-enabled device (optional) – for coordinates Bag or backpack (optional) – to hold samples Paper towels (optional) 2 oz plastic cup (any small cup or container will do, but I prefer this size) Pipette or eye-dropper (optional) Spring water (tardigrades prefer this, but any water should be fine for short-term use) Foldscope When I’ve found the lichen that I would like to collect, I take out a paper bag, open it up, and then take out my razor blade.
Here are the materials you will need:
Paper bags Razor blade or pocket knife (optional) Pen or Marker GPS-enabled device (optional) – for coordinates Bag or backpack (optional) – to hold samples Paper towels (optional) 2 oz plastic cup (any small cup or container will do, but I prefer this size) Pipette or eye-dropper (optional) Spring water (tardigrades prefer this, but any water should be fine for short-term use) Foldscope When I’ve found the lichen that I would like to collect, I take out a paper bag, open it up, and then take out my razor blade.

View in Media Gallery
I position the paper bag below the lichen, then begin to scrape the lichen into the bag using the razor blade. Keep the blade nearly flat, and try to lift the lichen off of the surface rather than crushing it up. The bag should catch any small pieces that happen to break off.



Pick up any large chunks and place them in the bag.


I will usually scrape the entirety of a small colony, but I always remember to respect the lichen, and take only what I will use. Some lichen colonies grow only a few mm per year, and can be many decades old.

View in Media Gallery
The lichen from these two small colonies should be enough for a couple samples that I can use many times over.

View in Media Gallery
I close the bag, and fold it such that I have space to write down information about my sample.

View in Media Gallery
I now take out my marker and write down my initials, the date on which the sample was collected, and the sample number (not shown here).

View in Media Gallery
I next write a description of the location that I took the sample from. For instance, my sample was taken from a wooden fence at the Governor’s Corner dormitories at Stanford University in California.

View in Media Gallery
If you have access to a GPS-enabled device, it is very useful to write down the coordinates of the location from which you collected the sample. By doing this, you can always return to the spot if you forget where it is, or even tell other people exactly where to find the original location of your sample.
I use a compass application on my smartphone to determine the coordinates of my sample locations.
I use a compass application on my smartphone to determine the coordinates of my sample locations.


Last, I write down a description of the sample itself. If I knew more about lichen taxonomy, I would make a guess at the genus and/or species, but I usually settle for a description of the color and/or texture.

View in Media Gallery
The same process also works for mosses on trees, rocks, dirt, etc. There was a tree nearby with a lot of moss at its base, so I decided to collect samples there as well.


First I took a sample of the moss that grew on the tree itself.

View in Media Gallery
I scraped chunks of the moss from the tree using my razor blade and placed them in a new paper bag.

View in Media Gallery
With this particular sample, I found it easier to pull off the moss with my hands.


After placing this moss in the paper bag, I next moved to the moss growing on the dirt around the tree.

View in Media Gallery
Again, I’m no expert at identifying moss taxa, but from my untrained perspective this looked like Bryum argentum , which has been reported to house a species of tardigrade called Milnesium tardigradum . The silver tips and conical shape of the moss led me to this guess.

View in Media Gallery
I used the razor blade to separate this moss from the ground, taking as little dirt as possible. Although tardigrades could be in the soil, it is much more likely that they are within the pockets of the moss, and too much dirt in the sample will make it difficult to search through later on.

View in Media Gallery
Once the chunks were free from the dirt, I collected them with my hands and put them in the paper bag that already contained moss from the tree.

View in Media Gallery
I labeled the bag as I did for the lichen, writing my name, the date, sample number, location description, coordinates, and sample description. I stored this in the plastic bag with my lichen sample, and headed back to the lab.

View in Media Gallery
Once I arrived at the lab, I found a drawer which could serve as a cool, dry place that I could store my samples in.
Please note that you do not need access to a laboratory or any laboratory equipment for finding tardigrades.
Please note that you do not need access to a laboratory or any laboratory equipment for finding tardigrades.

View in Media Gallery
If you won’t be using your samples right away, store them in a cool, dry place like this drawer until you need them. This will allow them to dry out slowly before use.

View in Media Gallery
Since I planned to use my samples right away, I didn’t actually bother storing them.
When tardigrades are taken from dry moss and lichen, they are most likely in their cryptobiotic “tun” state. This means that you will have to “rehydrate” the sample, soaking it in water for at least 4-8 hours before you can search it for tardigrades. Once the tardigrades are introduced to water, they will begin to enter their “active” state, leaving the pockets of the moss/lichen that they originally inhabited and swimming out into the water to find food. From this water, the tardigrades can be collected and imaged. I usually start the rehydration process the day before I intend to search for tardigrades in my samples.
First, I lay out a paper towel on a flat surface for each sample I plan to rehydrate. This isn’t absolutely necessary, but helps in the case of spills or wet containers.
When tardigrades are taken from dry moss and lichen, they are most likely in their cryptobiotic “tun” state. This means that you will have to “rehydrate” the sample, soaking it in water for at least 4-8 hours before you can search it for tardigrades. Once the tardigrades are introduced to water, they will begin to enter their “active” state, leaving the pockets of the moss/lichen that they originally inhabited and swimming out into the water to find food. From this water, the tardigrades can be collected and imaged. I usually start the rehydration process the day before I intend to search for tardigrades in my samples.
First, I lay out a paper towel on a flat surface for each sample I plan to rehydrate. This isn’t absolutely necessary, but helps in the case of spills or wet containers.

View in Media Gallery
I set out my sample bags on the corner of each paper towel so that I know which sample I’m handling at all times.

View in Media Gallery
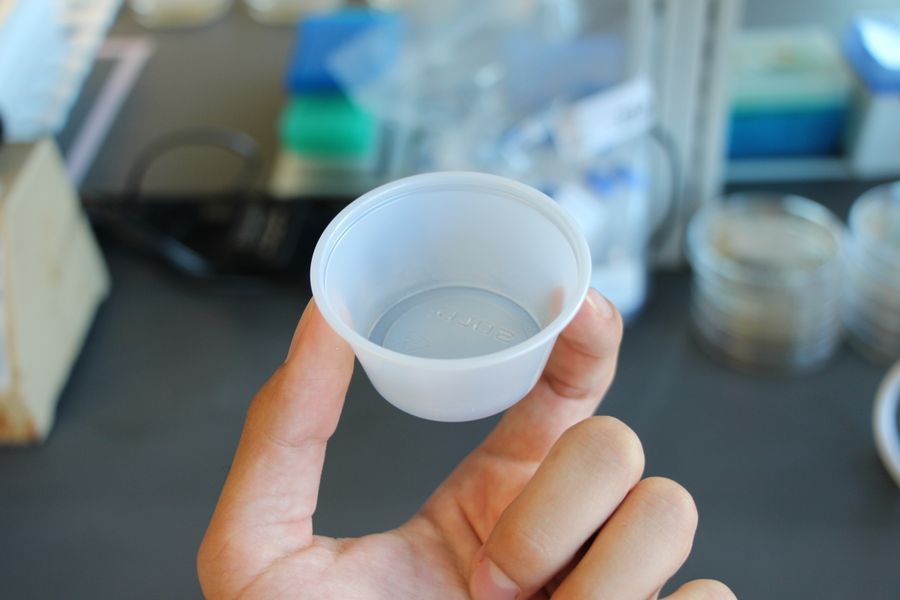
I place a 2 oz plastic cup on each paper towel that I will use for rehydrating the samples. You can use any small container that you have access to.

View in Media Gallery
I only use these particular plastic cups because my samples tend to fit well within them, and I don’t have to use too much water to completely soak the samples (more water means more to search through).
View in Media Gallery
Next I set a pipette on each paper towel for future use in transferring water after rehydration. I use glass Pasteur pipettes with a rubber bulb, but any pipette or eye-dropper should work fine. If you don’t have access to these items, you could also try pouring water after rehydration, using a straw, etc.


Once everything is in place, open up your sample bags, and remove their contents.


Place your collected sample face-down within the plastic cups. This helps to better soak the parts of the moss and lichen that are likely to house tardigrades.

View in Media Gallery
Repeat this with all other samples you plan to use.


If the cup becomes too full to allow everything to soak properly within a small volume of water, grab another cup and transfer some of your sample, dividing the contents so that they will fit.


I now take a bottle of spring water and pour out about 20 ml for each sample. It’s fine to do this without measuring the volume of water that you’re using. Just use as much as it takes to comfortably soak your samples.



I pour the water into each sample cup.

View in Media Gallery
To make sure that my samples are sufficiently soaked, I use a pipette to submerge them in the water. Again, you don’t need a pipette. You can do this with a stick, your finger, etc.


All that’s left to do is wait. As I mentioned, I usually let these soak overnight, but I’ve had success finding some active tardigrades in as little as 4 hours after soaking.

View in Media Gallery
When I return to my samples the next day, I usually find that some water has evaporated. This is fine, so long as there is still some water remaining in the cup. If all of it evaporates, your tardigrades will return to their “tun” state, and you will need to repeat the rehydration process.

View in Media Gallery
Sometimes moss samples will also absorb the water that they were soaked in. When this happens, I squeeze my moss like a sponge to release its water into the cup, then discard the moss in a separate container. The tardigrades will be happily swimming in the water that you squeezed out, so you don’t have to worry too much about them lingering around in the moss that you set aside.

View in Media Gallery
I now prepare my Foldscope slides, placing a cover slip on only one side in preparation for a water sample wet mount.


I take my pipette and attach the rubber bulb, then collect some of the water from my soaked samples.
View in Media Gallery
I like to take water from the edges of the cups, since I’ve found that tardigrades often crawl among the small pieces of debris that settle there.


I place a drop of the water on the sticky side of the slide, then place another cover slip on top of the slide to encapsulate my sample. I usually use my fingernail to press down the edges of the coverslips and ensure that no water escapes, creating a sealed pocket of water on the slide.

View in Media Gallery
The slides are now ready for viewing with the Foldscope!

View in Media Gallery
I use this flat, movable lamp as a uniform source of transmitted light (no light module), similar to the methods in Manu’s recent post on focus-locking and ambient light .

View in Media Gallery
I do a quick scan around the sample and look for tardigrades. If you don’t find them right away, don’t worry. Just put a new drop of water from your samples on the slide and continue your search.

View in Media Gallery
I found the first tardigrade of many after searching through five drops of water from the lichen sample taken off of the wooden fence. You can use my images and videos as a reference for what you should be looking for.

View in Media Gallery
Take a look at what I found!
I used Manu’s angular illumination technique to alter the contrast of my images and get a some different views of the tardigrades.
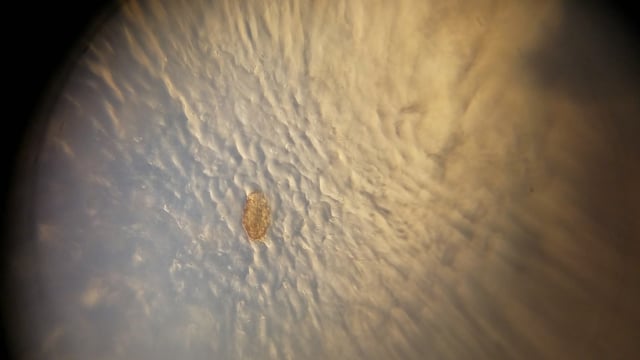
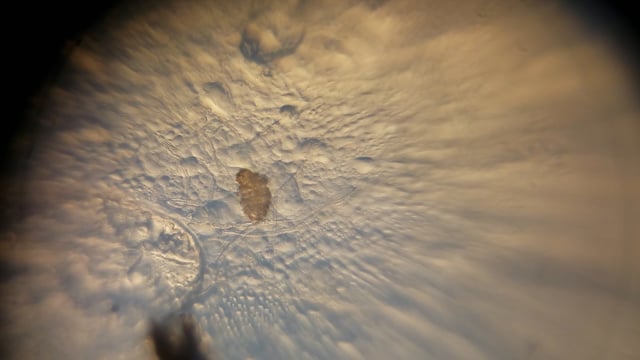
Lastly, I achieved what I call an “aquarium” view of the tardigrades by sealing off a large droplet of water near the edge of a wet mount slide, and then viewing it using angular illumination. As you’ll see, it looks like the tardigrade is in a tiny aquarium!
I hope you found this guide helpful, and I wish you the happiest of times searching for these amazing creatures around the globe.
I’m very excited to hear your answers to the following questions:
What do the tardigrades in your area look like? What did you find them on? What other creatures were in the water with them?
For more info on tardigrade biology and working with tardigrades, please see the following resource:
http://tardigraderesearch.blogspot.com/
This post is open to read and review on The Winnower.
I’m very excited to hear your answers to the following questions:
What do the tardigrades in your area look like? What did you find them on? What other creatures were in the water with them?
For more info on tardigrade biology and working with tardigrades, please see the following resource:
http://tardigraderesearch.blogspot.com/
This post is open to read and review on The Winnower.
Sign in to commentNobody has commented yet... Share your thoughts with the author and start the discussion!
More Posts from Ricky Cordova
No more posts from this author.