Microfluidics: a primer
 Aug 14, 2016 • 2:51 PM UTC
Aug 14, 2016 • 2:51 PM UTC United States
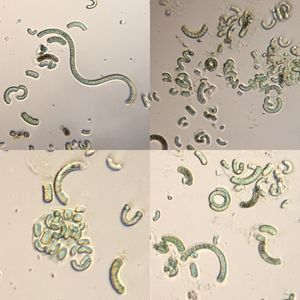
United States 140x Magnification
140x Magnification Plants
Plants
Damon Tighe
I currently work as a Curriculum and Training Specialist at Bio-Rad laboratories and help teachers incorporate more biotechnology into their classroom. I worked in DNA sequencing for years including the Human Genome Project and single cell genomics (termite, cow, and other guts). I have a love of the outdoors that I spread to the public via Calnature.org. I enjoy backpacking, picking mushrooms and photography.
15posts
17comments
9locations
View in Media Gallery
This post is going up a little before I’d like, in that I’ve made a few steps towards a tool for the Foldscope, but the tool isn’t easy to use or refined yet. A teacher I work with outside of Denver, Colorado that does an amazing job of letting students really taste whats its like to work in science by having students take on a research project for a large chunk of the year with paper research, proposals, meetings, working with mentors and troubleshooting, followed by troubleshooting and occasionally pivoting has a group of students with a little more of a biotech meets engineering lean this year and I want to document where I am at on this project so that they can hopefully blow it out of the water. Most of the material below is from May/June and I just haven’t had time to pick up and move forward.
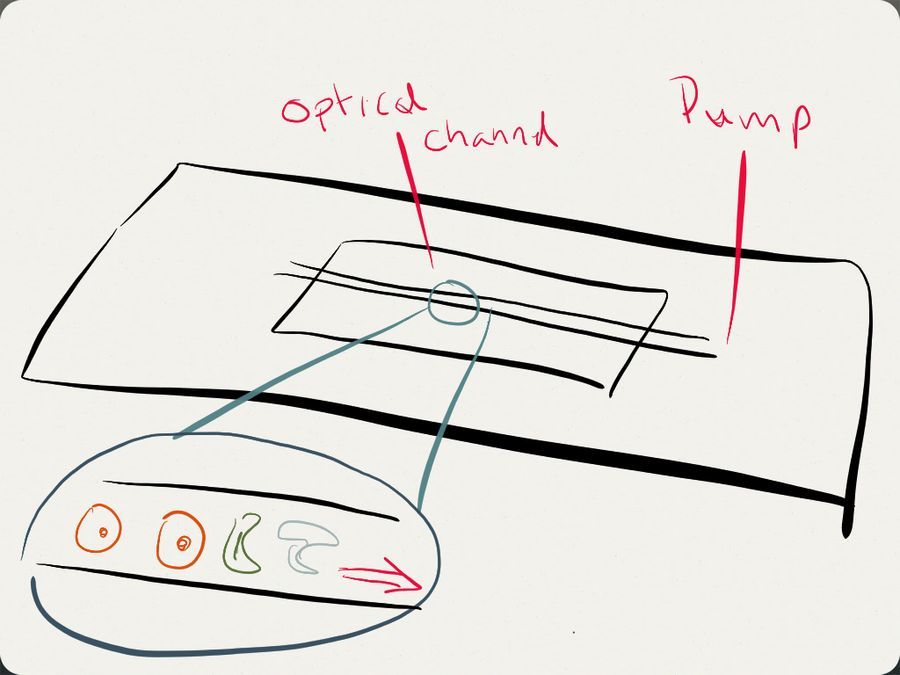
The Goal: I’m interested in building some cheap microfluidic devices that can either be reused or could be easily built in the field with a few supplies. The most basic device idea is just a simple channel that would be wide enough for red blood cells, diatoms and other cells to be flowed in front of the cell phone mounted to the Foldscope. The hope is that it could greatly reduce the time that is needed to scan a slide to look for cell types of interest as one could flow the cells past the lens and just have the cell phone capturing video, which may allow for cell counting and maybe with a little bit of image processing even identifying cell types. I would prefer the device be able to run without electricity, maybe using capillary action or something similar as the “pump” to move volumes through the channel.
The Goal: I’m interested in building some cheap microfluidic devices that can either be reused or could be easily built in the field with a few supplies. The most basic device idea is just a simple channel that would be wide enough for red blood cells, diatoms and other cells to be flowed in front of the cell phone mounted to the Foldscope. The hope is that it could greatly reduce the time that is needed to scan a slide to look for cell types of interest as one could flow the cells past the lens and just have the cell phone capturing video, which may allow for cell counting and maybe with a little bit of image processing even identifying cell types. I would prefer the device be able to run without electricity, maybe using capillary action or something similar as the “pump” to move volumes through the channel.

View in Media Gallery
How to make a channel?
Etching? I tried just etching in a glass slide with a quartz crystal I had around except I found that my manual dexterity did not produce nice straight channels nor were the channels optically clear enough were I felt I could be able to resolve images afterwards. pseudo-Lithography? For me this ended up being the easiest route – pick a material that could be a nice sized channel, smash it between a glass slide and a cover slip with a material that could harden around it and then try to remove the channel material Channel Material – Human hair – I figured it was a commodity easy to come by, it happens to be relatively smooth so possible to remove from the fixing material once it has set. For this channel making application I’m lucky that my hair is only sort of curly so I was able to pull on the hair to make it straight across the slide for a good channel Fixing Material – Wax – I used just a candle I had around to first add two drops of hot wax to either side of the slide which allowed me to hold a piece of hair tight between them until the wax set and kept the hair straight. I then dripped wax on to the center of the slide over the hair and placed a cover slip over it, pressing down gently with a piece of cloth so as not to put fingerprints all over it. I let it set up for 30 minutes and then removed the hair. I didn’t image it on the foldscope because I was being lazy, but you can see the channel below is pretty rough. I tried a few iterations where I would slide the hair back and forth multiple times to make it smooth, but it was never as optically clear as I’d like.
Etching? I tried just etching in a glass slide with a quartz crystal I had around except I found that my manual dexterity did not produce nice straight channels nor were the channels optically clear enough were I felt I could be able to resolve images afterwards. pseudo-Lithography? For me this ended up being the easiest route – pick a material that could be a nice sized channel, smash it between a glass slide and a cover slip with a material that could harden around it and then try to remove the channel material Channel Material – Human hair – I figured it was a commodity easy to come by, it happens to be relatively smooth so possible to remove from the fixing material once it has set. For this channel making application I’m lucky that my hair is only sort of curly so I was able to pull on the hair to make it straight across the slide for a good channel Fixing Material – Wax – I used just a candle I had around to first add two drops of hot wax to either side of the slide which allowed me to hold a piece of hair tight between them until the wax set and kept the hair straight. I then dripped wax on to the center of the slide over the hair and placed a cover slip over it, pressing down gently with a piece of cloth so as not to put fingerprints all over it. I let it set up for 30 minutes and then removed the hair. I didn’t image it on the foldscope because I was being lazy, but you can see the channel below is pretty rough. I tried a few iterations where I would slide the hair back and forth multiple times to make it smooth, but it was never as optically clear as I’d like.



Fixing Material – Clear Nail Polish – Trying to think what material would be inexpensive and may give me a more optically clear and smooth channel I settled upon trying clear nail polish and it worked surprisingly well. The downsides are that it does seem to seal all the way and the channel can collapse overtime and the material would often generate little bubbles during the drying process that would then interfere with the channel. Below you can see I use hair straightened between wax droplets to get a channel and in the microscope image you can see the channel is relatively clear although a little collapsed because the nail polish doesn’t completely harden beneath the glass. I assume the organic solvent can’t escape keeping it slightly fluid


This is all just rough sketches at this point in time to hopefully get others here excited about not just going out and documenting the microcosmos but thinking of more tools that could go hand in hand with the foldscope to make it even easier to quickly explore the world with.
Sign in to commentNobody has commented yet... Share your thoughts with the author and start the discussion!

 0 Applause
0 Applause 0 Comments
0 Comments