Tips on Viewing Life in a Water Drop
 Dec 28, 2015 • 9:59 AM UTC
Dec 28, 2015 • 9:59 AM UTC Unknown Location
Unknown Location 140x Magnification
140x Magnification Microorganisms
Microorganisms
Aatish Bhatia
Learn about the author...
6posts
3comments
1locations
View in Media Gallery
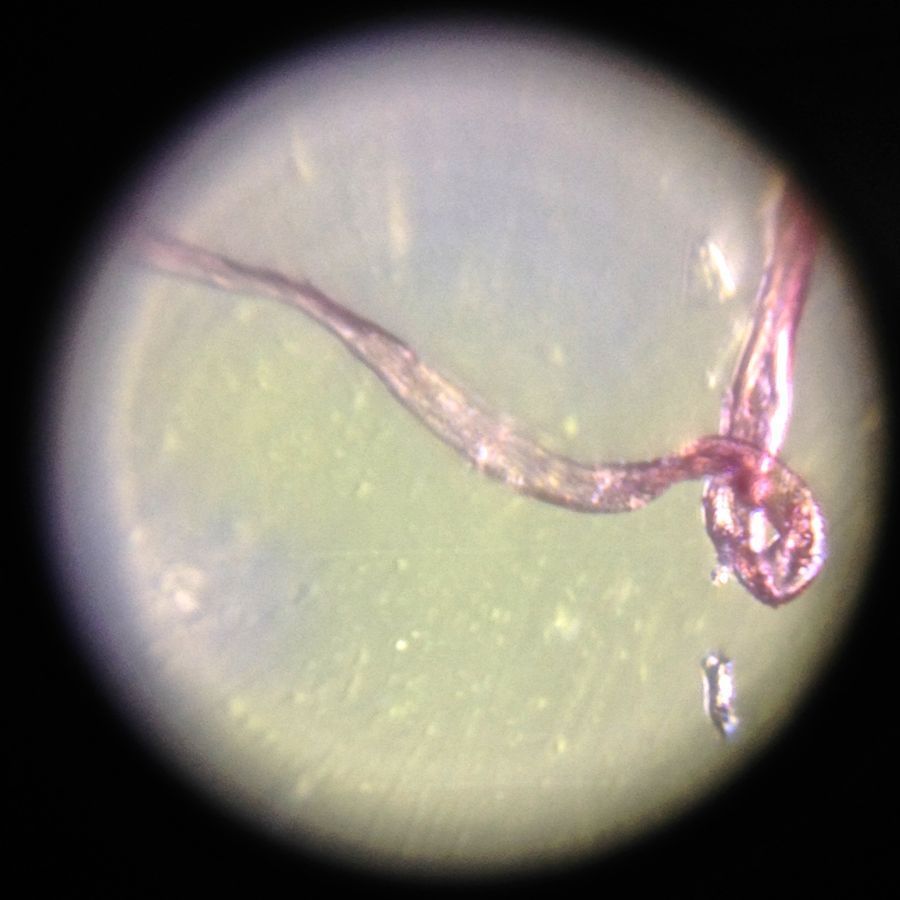
I’ve been trying to image life in a water drop under the foldscope, and found it trickier than I expected. Here’s a video of a recent attempt.
I don’t recognize any of the things I’m looking at, so if anything looks familiar to you, please drop a note in the comments.
Anyway, I reached out to Manu for some pointers on imaging life in water. I’m pasting below his response.
Really nice 🙂 Water from a pond has almost an unbounded diversity of life; once you get a hang of how to make slides – it’s really incredible. For an example – see this post : https://microcosmos.foldscope.com/?p=3500
Tips:
1) I think you are using the 450x lens in that video; a lower mag larger field of the view 150x lens is a better starting choice.
2) Live water samples requires making slides well. Use a glass slide for best resolution. Put two pieces of tape on two side (double sided tape is provided in the kit); put cover slip on the top. This allows just enough space for the organisms to survive.
3) Collect a drop of water from someplace which has a slightly “green” color – due to some algae growth. That’s the best indication that the water is not so polluted that it kills everything – frankly; anytime I have sampled water – it’s always got an incredible diversity.
Looking forward to trying these tips out!
Anyway, I reached out to Manu for some pointers on imaging life in water. I’m pasting below his response.
Really nice 🙂 Water from a pond has almost an unbounded diversity of life; once you get a hang of how to make slides – it’s really incredible. For an example – see this post : https://microcosmos.foldscope.com/?p=3500
Tips:
1) I think you are using the 450x lens in that video; a lower mag larger field of the view 150x lens is a better starting choice.
2) Live water samples requires making slides well. Use a glass slide for best resolution. Put two pieces of tape on two side (double sided tape is provided in the kit); put cover slip on the top. This allows just enough space for the organisms to survive.
3) Collect a drop of water from someplace which has a slightly “green” color – due to some algae growth. That’s the best indication that the water is not so polluted that it kills everything – frankly; anytime I have sampled water – it’s always got an incredible diversity.
Looking forward to trying these tips out!
Sign in to commentNobody has commented yet... Share your thoughts with the author and start the discussion!

 0 Applause
0 Applause 0 Comments
0 Comments


