Beauty of Kitchen Sink Experiments
 Jan 22, 2015 • 8:41 AM UTC
Jan 22, 2015 • 8:41 AM UTC Unknown Location
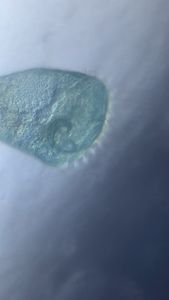
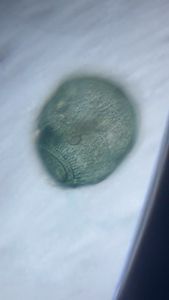
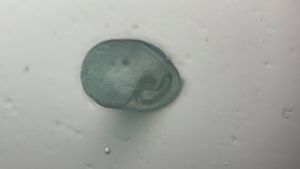
Unknown Location 140x Magnification
140x Magnification Microorganisms
Microorganisms
Manu Prakash
I am a faculty at Stanford and run the Prakash Lab at Department of Bioengineering at Stanford University. Foldscope community is at the heart of our Frugal Science movement - and I can not tell you how proud I am of this community and grassroots movement. Find our work here: http://prakashlab.stanford.edu
266posts
1198comments
42locations

View in Media Gallery
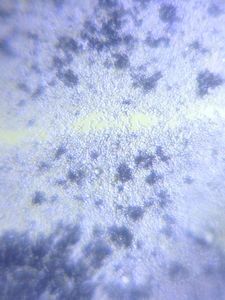
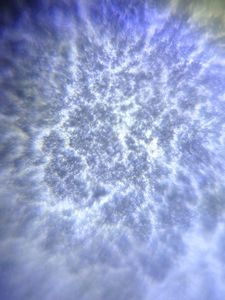
I strongly believe “kitchen sink” should be the heart of science activity in any house hold. Just think about it; 100’s of fluid mechanics problems when a jet hits a flat sheet. You add soap to it, and loads of foam problems to think about. And now, since you have a microscope, you can add tons of biological problems to it. So this post is about – what’s growing in my kitchen.
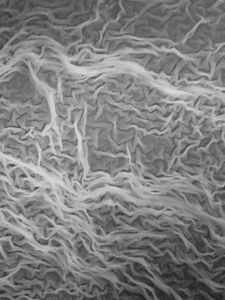
Just from last night kitchen cleaning, I discovered a beautiful single layer thick cell sheet (probably a bacilus bacterial interfacial biofilm – but j need to confirm it) that I imaged with my foldscope. It was beautiful!!
Some observations
1. The film truly was single cell later thick.
2. The patterns arise from buckling of a thin sheet. Imagine a sheet that is growing but only has a fixed area. What does it do – it wrinkles.
3. I saw a “black” substance in the films; which was only present in parts that were intact. I am really curious about that.. It could be a secreted compound that gives an irrescident feel to the film. I only found it in intact films.
4. At the edges, they easily break into single cells.
I head is spinning with so many questions. Can’t even begin to write them down. Are these films hydrophobic. Are they hydrophobic on one side, and Philic on the other. What about water underneath. Do they cut evaporation completely?
I am just mesmerized by the beauty of the microscopic world. Another reason to not clean my kitchen (or not clean it so frequently).
Just from last night kitchen cleaning, I discovered a beautiful single layer thick cell sheet (probably a bacilus bacterial interfacial biofilm – but j need to confirm it) that I imaged with my foldscope. It was beautiful!!
Some observations
1. The film truly was single cell later thick.
2. The patterns arise from buckling of a thin sheet. Imagine a sheet that is growing but only has a fixed area. What does it do – it wrinkles.
3. I saw a “black” substance in the films; which was only present in parts that were intact. I am really curious about that.. It could be a secreted compound that gives an irrescident feel to the film. I only found it in intact films.
4. At the edges, they easily break into single cells.
I head is spinning with so many questions. Can’t even begin to write them down. Are these films hydrophobic. Are they hydrophobic on one side, and Philic on the other. What about water underneath. Do they cut evaporation completely?
I am just mesmerized by the beauty of the microscopic world. Another reason to not clean my kitchen (or not clean it so frequently).






Sign in to commentNobody has commented yet... Share your thoughts with the author and start the discussion!

 0 Applause
0 Applause 0 Comments
0 Comments