Silver Crystals Using Tollens’ Test Reagents
 Nov 07, 2015 • 1:32 AM UTC
Nov 07, 2015 • 1:32 AM UTC Unknown Location
Unknown Location 140x Magnification
140x Magnification Non-Living
Non-Living
Marie Herring
Learn about the author...
4posts
13comments
1locations


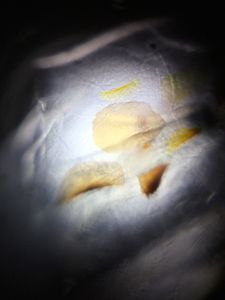
Have you ever seen the Silver Mirror trick at a chemistry show? That’s when silver is precipitated out of solution onto a glass surface. The demo consists of a series of clear solutions being combined in a glass flask. As the presenter swirls the flask, a shiny mirror forms on the inner surface of the glass. The reaction utilizes Tollens’ reagent, which is used to determine the difference between aldehydes and ketones. I thought it might be cool to modify the procedure slightly to be able to grow silver crystals. This turned out to be slightly more challenging than I predicted, mostly because the Tollen’s reagent is designed to form a smooth silver film on any glass surface, so crystals are not the first structures produced. However, with some tweaking and dilution, I was able to get a slide with some individual crystals. Above is a picture through the Foldscope and below is a macro photo of the prepared slide.

View in Media Gallery
Sign in to commentNobody has commented yet... Share your thoughts with the author and start the discussion!

 0 Applause
0 Applause 0 Comments
0 Comments