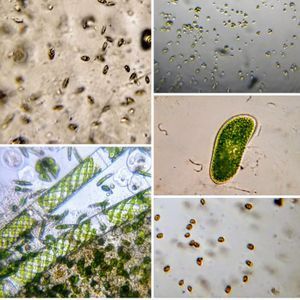
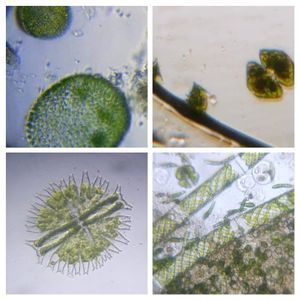
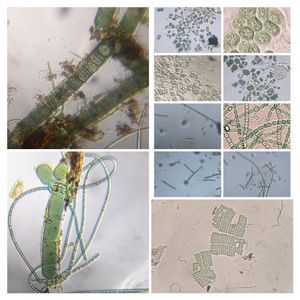
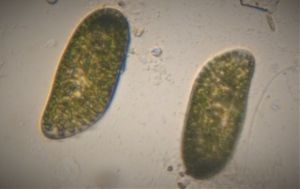
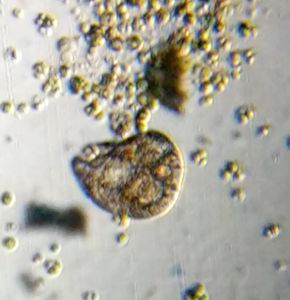
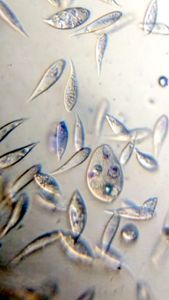
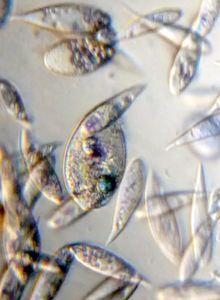
Bubbles in Saliva
 Feb 26, 2016 • 4:14 PM UTC
Feb 26, 2016 • 4:14 PM UTC Unknown Location
Unknown Location 140x Magnification
140x Magnification Microorganisms
Microorganisms
Laks Iyer
Human observer of life. https://sukshmadarshin.wordpress.com
97posts
1255comments
5locations

View in Media Gallery
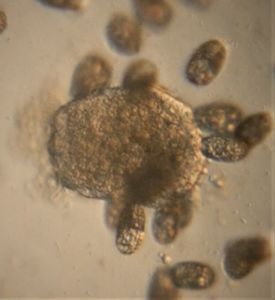
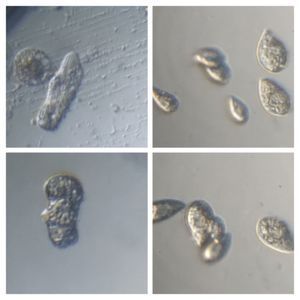
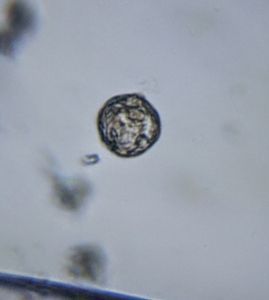
In the potato staining experiments , Aditi (DC-Micronaut) wondered what would happen if we spit on the potato, would we see the amyloplasts being digested? More on those experiments in a different post. What caught our attention were the salivary bubbles. You could see bubbles through bubbles and it made for a dainty sight. Coming to think of it, why should Saliva bubble?
Saliva is mainly composed of water (99.5%), the remaining 0.5% is made up of glycoproteins (proteins linked to sugars), digestive enzymes like amylases and lipases, protective proteins such as lysozyme,peroxidin, lactoferrin and immunoglobulins that kill bacteria, carbonic anyhydrase that has a role in taste physiology and various ions that maintain a pH balance between 6.2 and 7.4. The term “licking your wound”s is based on the observation that animals (humans included) are often seen licking their wounds and experiments have shown the saliva of various animals to possess antibacterial molecules that disinfect wounds. Saliva is extensively used across vertebrates for distinct purposes and it is fascinating to read.
One thing that proteins in solution do is to froth, e.g. Milk Froth. So the bubbling is likely to be due to the proteins in saliva. But does the type of protein in solution influence the type of the bubble? Could we detect secretory differences between humans by just studying the stability of salivary bubbles? Are the salivary enzymes better off on a bubble surface as compared to being suspended in a more watery interface? Never thought about bubbles as much until we saw this. Perhaps the experts on our forum have interesting answers.
-Aditi and Laks
Saliva is mainly composed of water (99.5%), the remaining 0.5% is made up of glycoproteins (proteins linked to sugars), digestive enzymes like amylases and lipases, protective proteins such as lysozyme,peroxidin, lactoferrin and immunoglobulins that kill bacteria, carbonic anyhydrase that has a role in taste physiology and various ions that maintain a pH balance between 6.2 and 7.4. The term “licking your wound”s is based on the observation that animals (humans included) are often seen licking their wounds and experiments have shown the saliva of various animals to possess antibacterial molecules that disinfect wounds. Saliva is extensively used across vertebrates for distinct purposes and it is fascinating to read.
One thing that proteins in solution do is to froth, e.g. Milk Froth. So the bubbling is likely to be due to the proteins in saliva. But does the type of protein in solution influence the type of the bubble? Could we detect secretory differences between humans by just studying the stability of salivary bubbles? Are the salivary enzymes better off on a bubble surface as compared to being suspended in a more watery interface? Never thought about bubbles as much until we saw this. Perhaps the experts on our forum have interesting answers.
-Aditi and Laks
Sign in to commentNobody has commented yet... Share your thoughts with the author and start the discussion!

 0 Applause
0 Applause 0 Comments
0 Comments_300x300.jpeg)