Dressing up protozoans (Vital Staining): Tetrahymena Vorax Part 2
 May 25, 2018 • 5:42 AM UTC
May 25, 2018 • 5:42 AM UTC Unknown Location
Unknown Location 140x Magnification
140x Magnification Microorganisms
Microorganisms
Laks Iyer
Human observer of life. https://sukshmadarshin.wordpress.com
97posts
1255comments
5locations
View in Media Gallery
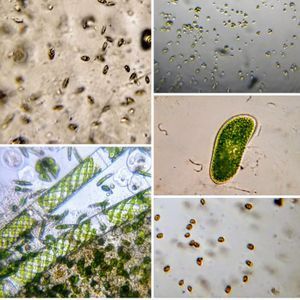
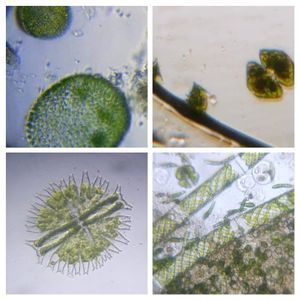
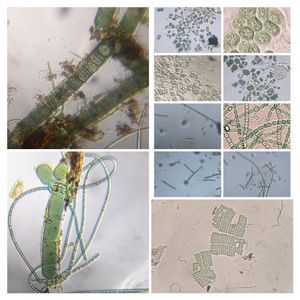
Have you ever tried to stain ciliates naively, i.e. without knowing any protocol, just adding methylene blue or some such dye? More than often all one sees are shriveled cells that don’t resemble anything like the live ones. For long, microscopists have used vital stains to stain ciliates live and I have always been wanting to try it. A while back I had purchased a set of vital stains for my home lab and with a culture of Tetrahymena vorax provided by the ASSETT program , I thought that this was a good time to test some of these stains. For vital stain concentrations, I used Richard Howey’s table that he provided in microscopy uk (one of my other favorite forums for amateur microscopy articles). http://www.microscopy-uk.org.uk/mag/indexmag.html?http://www.microscopy-uk.org.uk/mag/artfeb00/rhvital.html . For this I used Methylene Blue, Neutral Red, and Bismarck Brown. The final volume of the sample is about 20 microliters, and so I had to dilute my stock stains appropriately so that I add about 5 microliters to 15 microliters of culture. Without much ado, here are the results of my experiments.
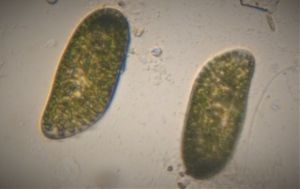
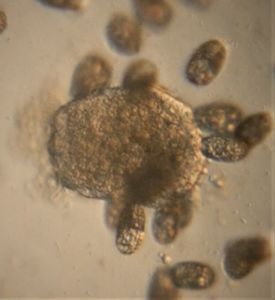
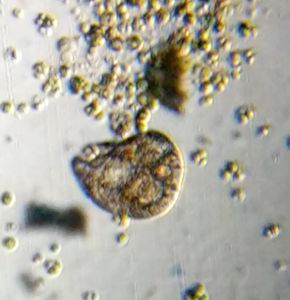
1. Methylene Blue normally stains nucleic acids and nuclei. Here it stained the macrostome form of T. vorax . In particular, it seems to stain the ciliates that have been engulfed along with the macronucleus
1. Methylene Blue normally stains nucleic acids and nuclei. Here it stained the macrostome form of T. vorax . In particular, it seems to stain the ciliates that have been engulfed along with the macronucleus
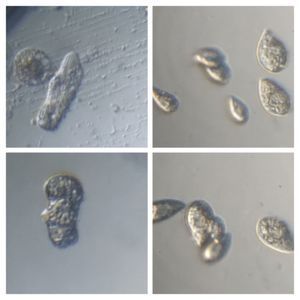
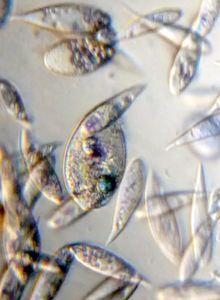
2. Neural red stains food vacuoles. The color of the vacuole depends on its acidity.
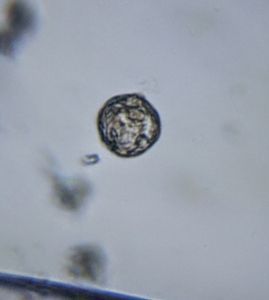
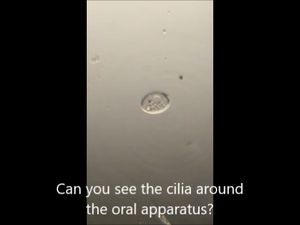
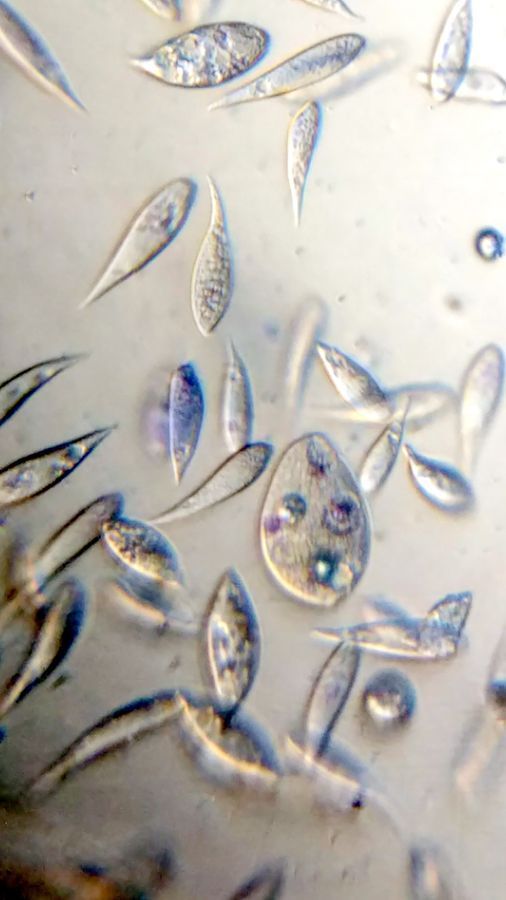
3. Bismarck Brown. I thought it provided a nice contrast for the cilia and ciliary bands. Can you see the developing macrostome?
I am going to experiment with increasing the concentration of the stains to see if it enhances staining in my future trials. In my next experiment, I shall describe an artificial ecosystem I created with T. vorax and Chlamydomonas , the results of which were fascinating.
This post is open to read and review on The Winnower.
This post is open to read and review on The Winnower.
Sign in to commentNobody has commented yet... Share your thoughts with the author and start the discussion!

 0 Applause
0 Applause 0 Comments
0 Comments_300x300.jpeg)